
The molecule also carries the important regulatory molecule nitric oxide bound to a globin protein thiol group, releasing it at the same time as oxygen. Hemoglobin is involved in the transport of other gases: It carries some of the body's respiratory carbon dioxide (about 20–25% of the total ) as carbaminohemoglobin, in which CO 2 is bound to the heme protein.

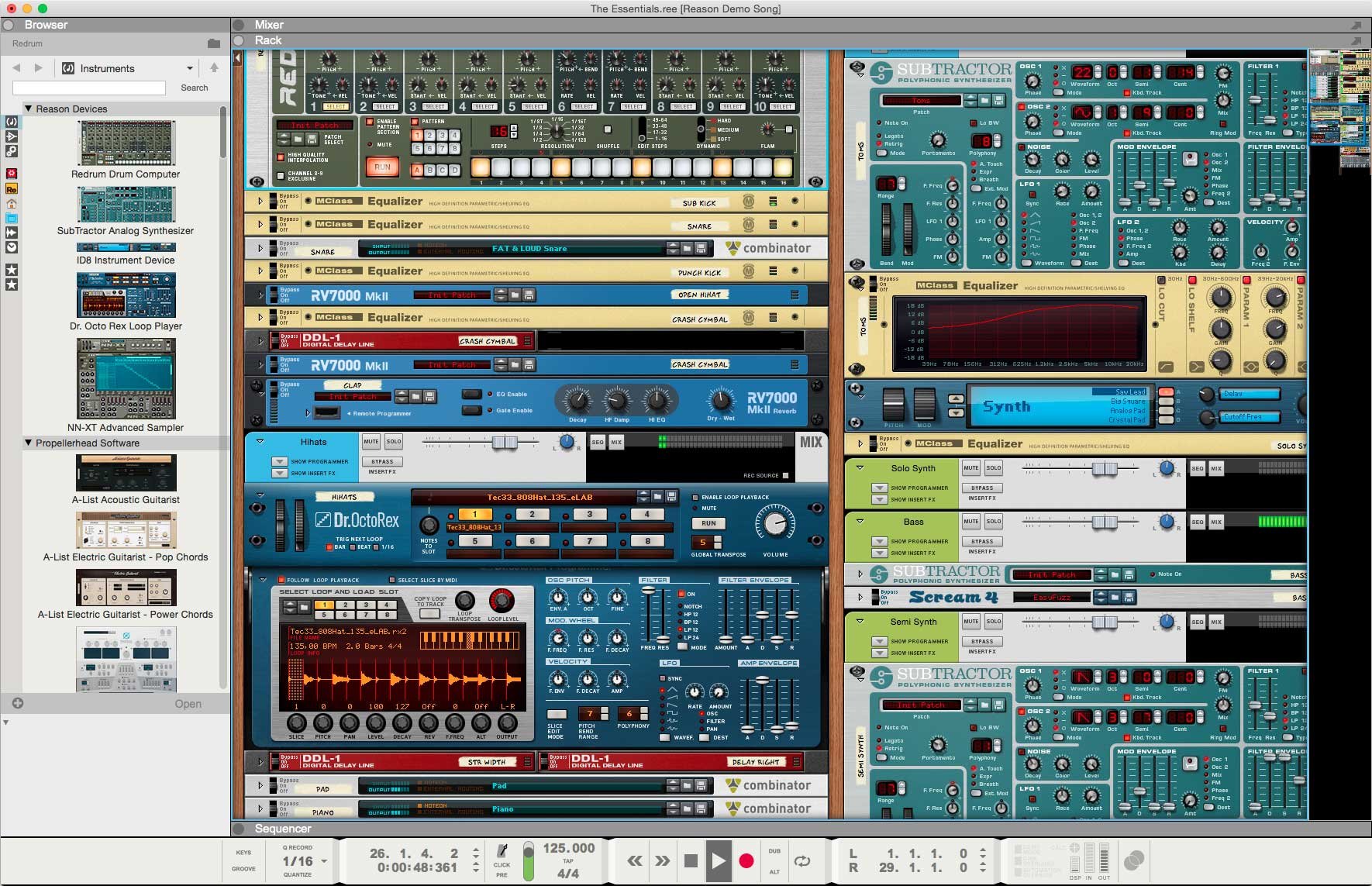
The mammalian hemoglobin molecule can bind (carry) up to four oxygen molecules. Hemoglobin has an oxygen-binding capacity of 1.34 mL O 2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood. In mammals, the protein makes up about 96% of the red blood cells' dry content (by weight), and around 35% of the total content (including water). A healthy individual has 12 to 20 grams of hemoglobin in every 100 mL of blood. There it releases the oxygen to permit aerobic respiration to provide energy to power the functions of the organism in the process called metabolism. Hemoglobin in blood carries oxygen from the lungs or gills to the rest of the body (i.e. Hemoglobin or haemoglobin ( spelling differences) (from the Greek word αἷμα, haîma 'blood' + Latin globus 'ball, sphere' + -in) ( / ˌ h iː m ə ˈ ɡ l oʊ b ɪ n, ˈ h ɛ m oʊ ˌ-/ ), abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein in the red blood cells (erythrocytes) of almost all vertebrates (the exception being the fish family Channichthyidae ) as well as the tissues of some invertebrates. α and β subunits are in red and blue, respectively, and the iron-containing heme groups in green.
